Most advanced tailored clinical trial service
HI-Mice service by Welala brings the most cost-effective clinical trial in drug discovery, medical research, and commercial purpose.
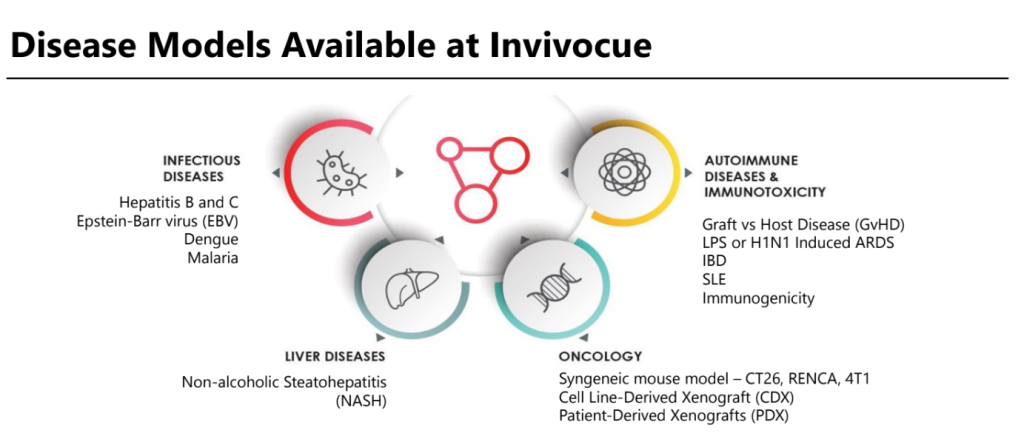
Most advanced tailored preclinical service
Welala’s HiMice services offer cutting-edge capabilities for advanced
preclinical research in drug discovery and medical studies.
with functional cell lineages, including T cells, B cells, NK cells, monocytes and dendritic cells.
meaning that these mouse models are suitable for long term studies of various immune and infectious diseases.






Contact our sales team for inquiry
Talk to our technical team to help to plan the objectives of your project.
Wait for 36-52 weeks depend on the purpose of your interest.
Our scienctific team will help you understand the results.
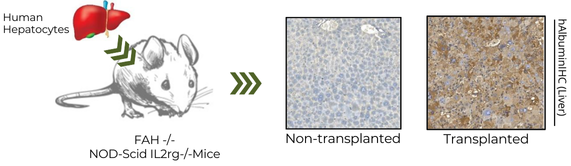
We use NIVO mice as the background strain. Like most NOD-Scid IL2Rγ-/- mice, NIVO are immunodeficient mice. The difference is that they have the entire IL2Rγ gene knocked-out in the NOD scid mice. There are non-functional mouse lymphoid cells (T, B cells), natural killer cells, dendritic cells and macrophages in NIVO mice. The mice are resistant to lymphoma development and are suitable for long-term studies. They can also support the engraftment and differentiation of CD34+ cells, PMBC, and cancer tumour tissue, and are suitable for studying human-specific disease mechanism
Nude mice are an early generation of immunodeficient mice with impaired T cells but partially defective B cells, NK, DC and Macrophage cells. Currently the most commonly used immunodeficient mouse model has two gene mutations (Prkdcscid and IL2rγ gene) in NOD mice, which improves the lifespan, immunodeficient and humanization capabilities of the mice. NIVO is an immunodeficient mouse model with Prkdcscid and IL2rγ gene mutations using the NOD background strain.
The life span of HiMice is 1 to 1.5 years.
The optimal window for experimental use is 12-20 weeks old.
We inject CD34+ HSC from human cord blood to immunodeficient mice. It takes 3-4 months for stem cells to develop into immune cells.
NK cells, T cells (T helper and T cytotoxic), B cells, monocytes and dendritic cells are expressed in HiMice.
HiMice will be tested for reconstitution of hCD45+ cells in the peripheral blood at 12-16 weeks post hematopoietic stem cells (HSC) transplantation using flow cytometry analysis. We calculate the percentage of human CD45 chimerism by dividing human CD45 events by the sum of human and mouse CD45 events. >20% of hCD45+ reconstitution in the peripheral blood is considered a good reconstitution.
The standard way to check for cell lineage reconstitution levels without sacrificing mice is by checking the peripheral blood. However, peripheral blood hCD45 reconstitution experiences fluctuations easily (based on circadian rhythms). The actual cell lineage is represented not only in the blood but also in the organs of HiMice. From our experience, hCD45 reconstitution in organs (e.g. spleen) is more reflective of functional immune cell populations in humans, and levels are usually consistently high (>80%) even with low peripheral blood reconstitution.
Reconstitution of certain human immune cells in humanized mice is often low due to the absence of important human cytokines which are essential for immune cell differentiation. Our proprietary technique of hydrodynamic injection of human cytokine plasmids is safe, effective and stably improves the human cytokine expression, as well as the reconstitution of human T, NK, dendritic cells and macrophages in HiMice.
The injection of cytokine plasmids is optional and is mainly based on the requirements of specific studies. Hydrodynamic Injection delivers the cytokine DNA when all the major cells have been developed, which optimizes the cytokine profiles of the cell populations without affecting their early development. (Transgenic or knock-in mice expressing human cytokines have been shown to develop anemia and loss of reconstitution due to exhaustion of HSCs from early differentiation. Those mice usually have shortened lifespans of 4-5 months after CD34+/PBMCs implantation.)
The cytokines can be customized with different combinations to meet specific needs. For example, if more macrophages are needed, then plasmids encoding M-CSF could be injected (similarly, IL-15 for NK cells, and GM-CSF and IL-3 for myeloid cells). For HiMice with a good repertoire of immune cells, a comprehensive combination of plasmids is used. Using the hydrodynamic Injection of human cytokine plasmids, HiMice with a diverse range of characteristics and applications can be made at minimal additional costs.
The effects of the cytokine boost (elevated numbers of specific immune cell populations) can last for 1 month.
In our models, we have 0-3% myeloid cell populations. We can use M-CSF to improve myeloid cell populations, or we can use GMCSF-IL3 to improve overall lineage reconstitution. [link to boosted reconstitution]
With the latter, the absolute number of myeloid cells will increase, even though its percentage will remain unchanged.
There is increasing evidence showing that human tumours change and evolve significantly in response to immune surveillance, which together with tumour-specific T cells and tumour antigens, are reflective of the in vivo human condition (Finn., O.J. J Immunol, 2018, 200 (2) 385-391).
The tumour responses to chemotherapy in humanized mice better mimic responses in the human in vivo environment (e.g. immune cell infiltration, exhaustion, and cytokine production) compared to the tumour responses in conventional mice models. Therefore, using HiMice for cancer studies generates data with higher translational value.
Besides chemotherapy studies, HiMice are also uniquely suited for immunotherapy studies, as they express the human immune system with functional cell lineages, which do not exist in conventional mice models.
Usually, cell-line derived xenografts (CDX) and patient-derived xenografts (PDX) can survive even with the complete human immune system present by developing immune-resistance (e.g. through immune checkpoints).
We highly recommend that clients first conduct a pilot study to confirm tumour growth in HiMice, as tumours are highly heterogenous.
Studies have shown that human T cells developed in humanized mice are tolerant to both human and mouse antigens, indicating efficient negative selection by both murine and human APCs (Traggiai et al. Science, 2004; 304:104–107). It has also been reported that mature human T cells from NOD/SCID/IL2rnull mice engrafted with human HSCs display cytotoxic functions in a human HLA-dependent manner, which indicates that at least some positive and/or negative selection of human T cells could occur in NOD/SCID/IL2rnull recipients (Ishikawa et al. Blood, 2005; 106: 1565-1573).
Extrathymic T cell maturation has been shown to occur in humans as well as nude mice in sites such as the liver and the intestines (Torfadottir et al. Clin. Exp. Immunol, 2006; 145:407-412; and Kennedy et al. J Immunol, 1992; 148: 1620-1629). Antigen presenting cells such as dendritic cells and macrophages can provide the human HLA to support T cell maturation at extrathymic sites. In a publication by our co-founder, it was shown that human antigen-specific adaptive immune responses can be enhanced by promoting CD11c+CD209+ human dendritic cell development in humanized mice (Chen et al. Journal of Immunology, 2012; 189:5223-5229).
Furthermore, the thymus in humanized mice is able to produce similar T cell subsets (e.g. CD4+/CD8+ double positive cells) as that of a human thymus. In a paper on using the humanised mouse model to test therapeutic drugs for hepatocellular carcinoma (Zhao et al. Gut, 2018; 67: 1845-1854), it was shown that T cells can respond to immunomodulatory drugs and drive disease progression, suggesting that T cells do respond well to disease and their immune function is not compromised in humanized mice.
No, a license is not required.
We can generate up to 100 mice from 1 donor. Larger quantities may be available upon specific request.
No. Our mice are housed in specific pathogen-free animal facilities and there are no additional husbandry requirements.
The lead time to generate HiMice is 3 to 4 months upon confirmed order. You may also check with us in advance to see if we have existing stock for your study requirements by sending your enquiry to .